
How to Use Lead Acid Battery: Examples, Pinouts, and Specs

 Design with Lead Acid Battery in Cirkit Designer
Design with Lead Acid Battery in Cirkit DesignerIntroduction
The lead acid battery is a rechargeable energy storage device that utilizes lead dioxide (PbO₂) as the positive electrode, sponge lead (Pb) as the negative electrode, and sulfuric acid (H₂SO₄) as the electrolyte. It is one of the oldest and most widely used battery technologies due to its reliability, cost-effectiveness, and ability to deliver high surge currents.
Common applications of lead acid batteries include:
- Automotive starting, lighting, and ignition (SLI) systems
- Uninterruptible power supplies (UPS) for backup power
- Renewable energy storage (e.g., solar and wind systems)
- Industrial equipment and forklifts
- Emergency lighting systems
Explore Projects Built with Lead Acid Battery
 Open Project in Cirkit Designer
Open Project in Cirkit Designer
 Open Project in Cirkit Designer
Open Project in Cirkit Designer
 Open Project in Cirkit Designer
Open Project in Cirkit Designer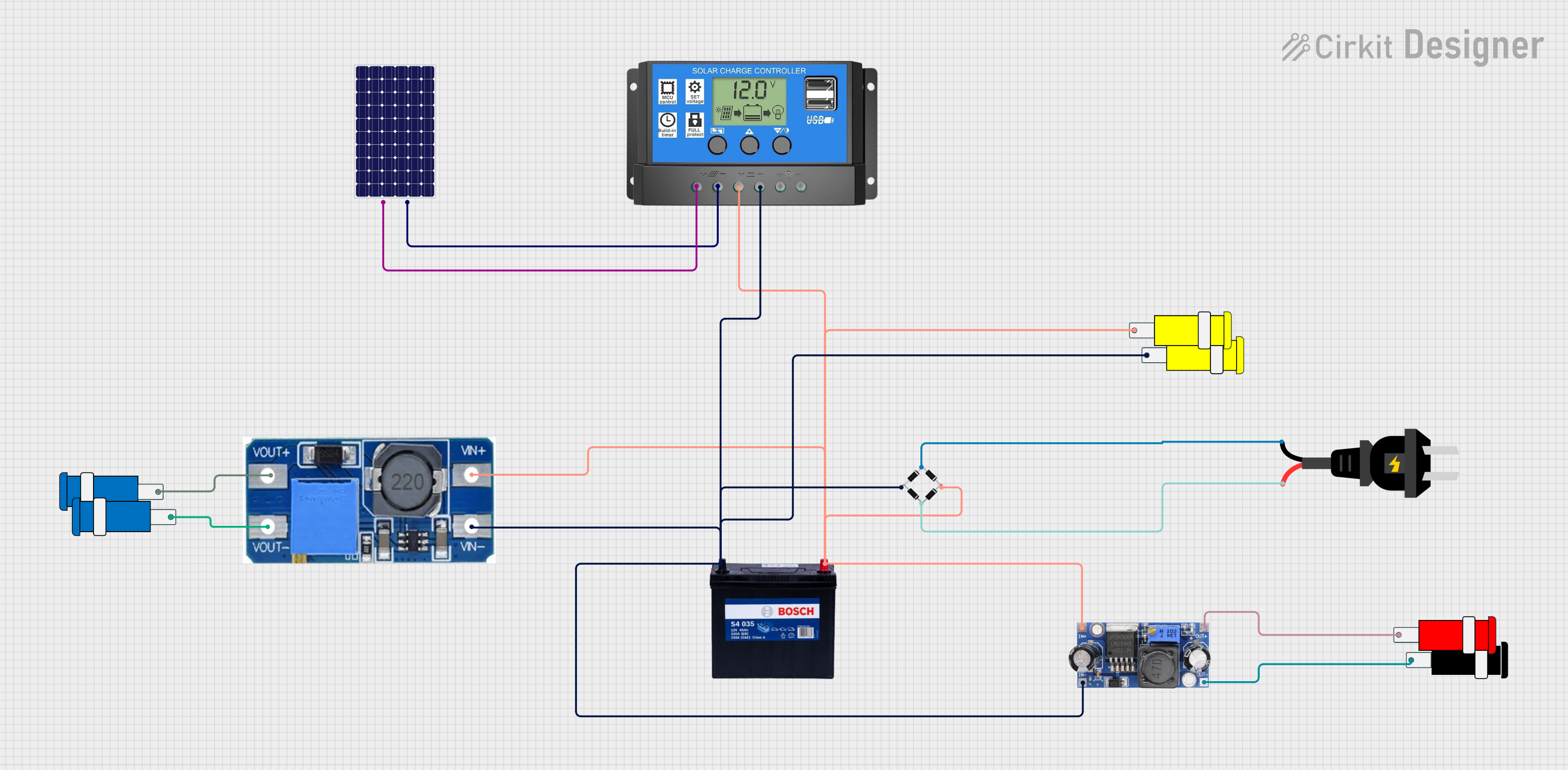
 Open Project in Cirkit Designer
Open Project in Cirkit DesignerExplore Projects Built with Lead Acid Battery

 Open Project in Cirkit Designer
Open Project in Cirkit Designer
 Open Project in Cirkit Designer
Open Project in Cirkit Designer
 Open Project in Cirkit Designer
Open Project in Cirkit Designer
 Open Project in Cirkit Designer
Open Project in Cirkit DesignerTechnical Specifications
Below are the key technical details of a typical lead acid battery. Note that specifications may vary depending on the specific model and manufacturer.
| Parameter | Value |
|---|---|
| Nominal Voltage | 2V per cell (commonly 6V or 12V for packs) |
| Capacity | 1.2Ah to 200Ah or more |
| Charge Voltage Range | 2.3V to 2.45V per cell |
| Discharge Voltage Range | 1.75V to 2.0V per cell |
| Maximum Discharge Current | Varies by model (e.g., 10A to 1000A) |
| Operating Temperature | -20°C to 50°C |
| Cycle Life | 200 to 1000 cycles (depending on usage) |
| Self-Discharge Rate | ~3% to 5% per month |
Pin Configuration and Descriptions
Lead acid batteries typically have two terminals:
| Terminal | Description |
|---|---|
| Positive (+) | Connects to the positive side of the circuit/load. |
| Negative (-) | Connects to the negative side of the circuit/load. |
Ensure proper polarity when connecting the battery to avoid damage to the battery or connected devices.
Usage Instructions
How to Use the Component in a Circuit
- Determine the Voltage and Capacity Requirements: Select a lead acid battery with the appropriate voltage (e.g., 6V or 12V) and capacity (Ah) for your application.
- Connect the Terminals:
- Connect the positive terminal of the battery to the positive side of the circuit.
- Connect the negative terminal of the battery to the negative side of the circuit.
- Charging the Battery:
- Use a compatible lead acid battery charger with the correct voltage and current ratings.
- Set the charger to the recommended charge voltage (e.g., 2.3V to 2.45V per cell).
- Avoid overcharging, as it can damage the battery and reduce its lifespan.
- Discharging the Battery:
- Ensure the load does not exceed the maximum discharge current rating.
- Avoid deep discharges below 1.75V per cell to prevent damage.
Important Considerations and Best Practices
- Ventilation: Lead acid batteries release hydrogen gas during charging. Use them in well-ventilated areas to prevent gas buildup.
- Temperature: Avoid exposing the battery to extreme temperatures, as this can affect performance and lifespan.
- Maintenance: For flooded lead acid batteries, periodically check and refill the electrolyte with distilled water as needed.
- Polarity: Always double-check the polarity before connecting the battery to a circuit to prevent short circuits or damage.
- Storage: Store the battery in a cool, dry place and recharge it periodically to prevent sulfation.
Example: Connecting a Lead Acid Battery to an Arduino UNO
To power an Arduino UNO with a 12V lead acid battery, you can connect the battery to the Arduino's VIN pin and GND pin. Below is an example code to read the battery voltage using an analog input pin.
// Example code to read lead acid battery voltage using Arduino UNO
const int batteryPin = A0; // Analog pin connected to battery voltage divider
const float voltageDividerRatio = 5.7; // Adjust based on resistor values used
const float referenceVoltage = 5.0; // Arduino's reference voltage (5V)
void setup() {
Serial.begin(9600); // Initialize serial communication
}
void loop() {
int analogValue = analogRead(batteryPin); // Read analog value from pin
float batteryVoltage = (analogValue / 1023.0) * referenceVoltage * voltageDividerRatio;
// Print the battery voltage to the Serial Monitor
Serial.print("Battery Voltage: ");
Serial.print(batteryVoltage);
Serial.println(" V");
delay(1000); // Wait for 1 second before the next reading
}
Note: Use a voltage divider circuit to step down the battery voltage to a safe range (0-5V) for the Arduino's analog input pin. Choose resistor values that provide the correct ratio for your battery voltage.
Troubleshooting and FAQs
Common Issues and Solutions
Battery Not Charging:
- Cause: Faulty charger or incorrect charge voltage.
- Solution: Verify the charger is functioning and set to the correct voltage.
Battery Drains Quickly:
- Cause: Over-discharge, sulfation, or aging battery.
- Solution: Avoid deep discharges and replace the battery if it has reached the end of its life.
Overheating During Charging:
- Cause: Overcharging or high ambient temperature.
- Solution: Use a charger with overcharge protection and ensure proper ventilation.
Low Voltage Output:
- Cause: Battery is discharged or damaged.
- Solution: Recharge the battery or replace it if it cannot hold a charge.
FAQs
Q: Can I use a lead acid battery in any orientation?
A: Sealed lead acid (SLA) batteries can be used in any orientation, but flooded lead acid batteries must remain upright to prevent electrolyte leakage.
Q: How do I dispose of a lead acid battery?
A: Lead acid batteries are hazardous waste and must be recycled at an authorized recycling facility. Do not dispose of them in regular trash.
Q: What is sulfation, and how can I prevent it?
A: Sulfation occurs when lead sulfate crystals form on the battery plates, reducing capacity. Prevent it by keeping the battery charged and avoiding prolonged storage in a discharged state.